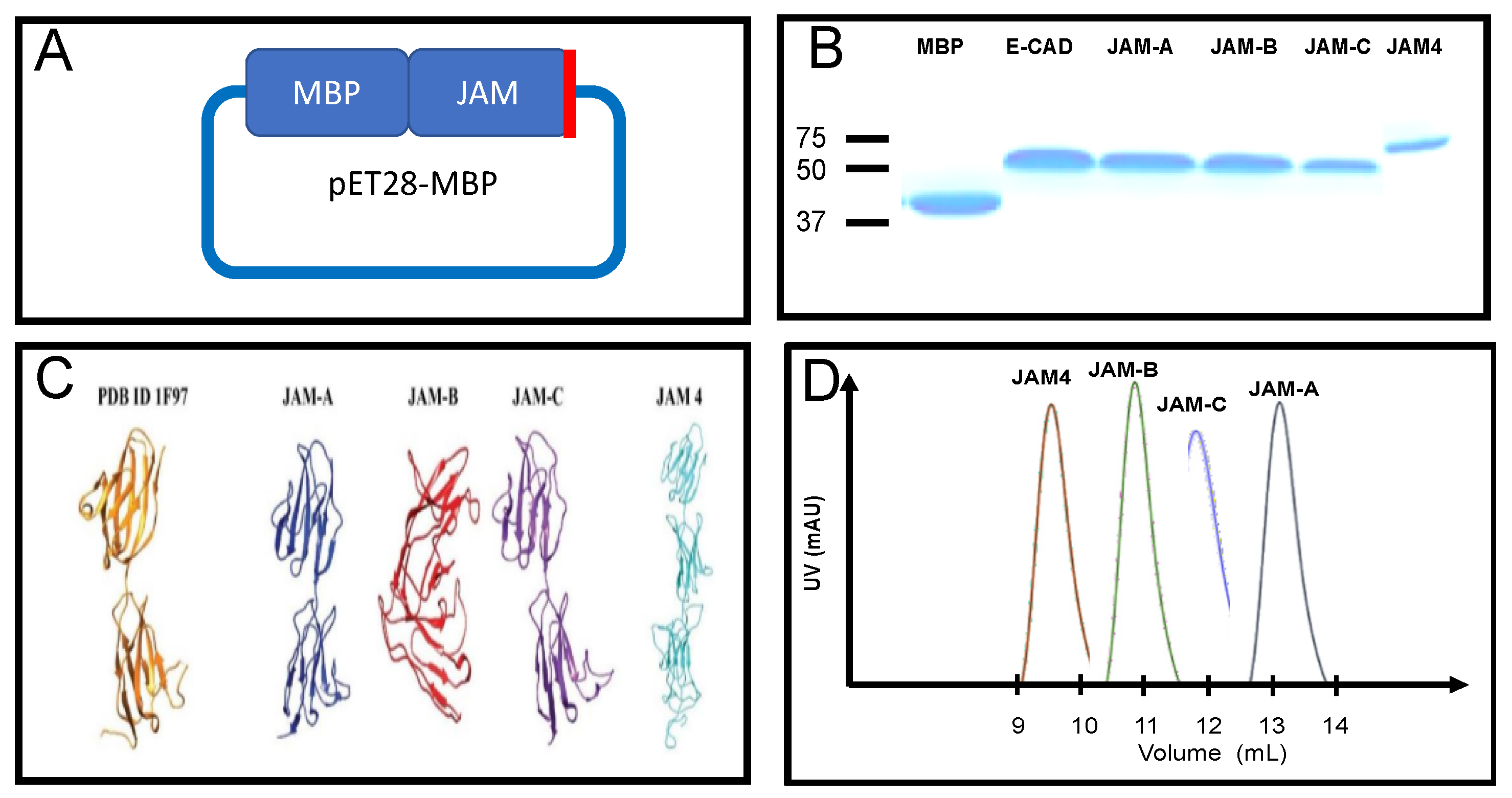
IJMS | Free Full-Text | Molecular Characterization of the Extracellular Domain of Human Junctional Adhesion Proteins
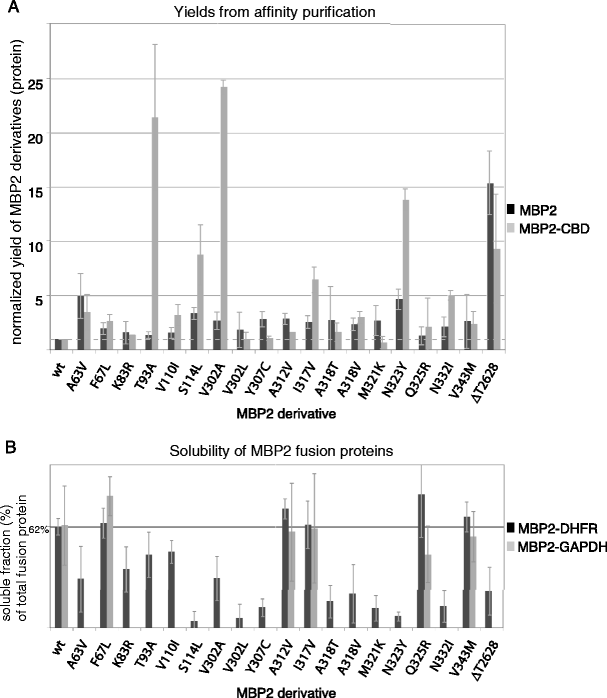
Mutations in maltose-binding protein that alter affinity and solubility properties | Applied Microbiology and Biotechnology

Mapping of the regions implicated in nuclear localization of multi‐functional DNA repair endonuclease XPF‐ERCC1 - Akahori - 2022 - Genes to Cells - Wiley Online Library
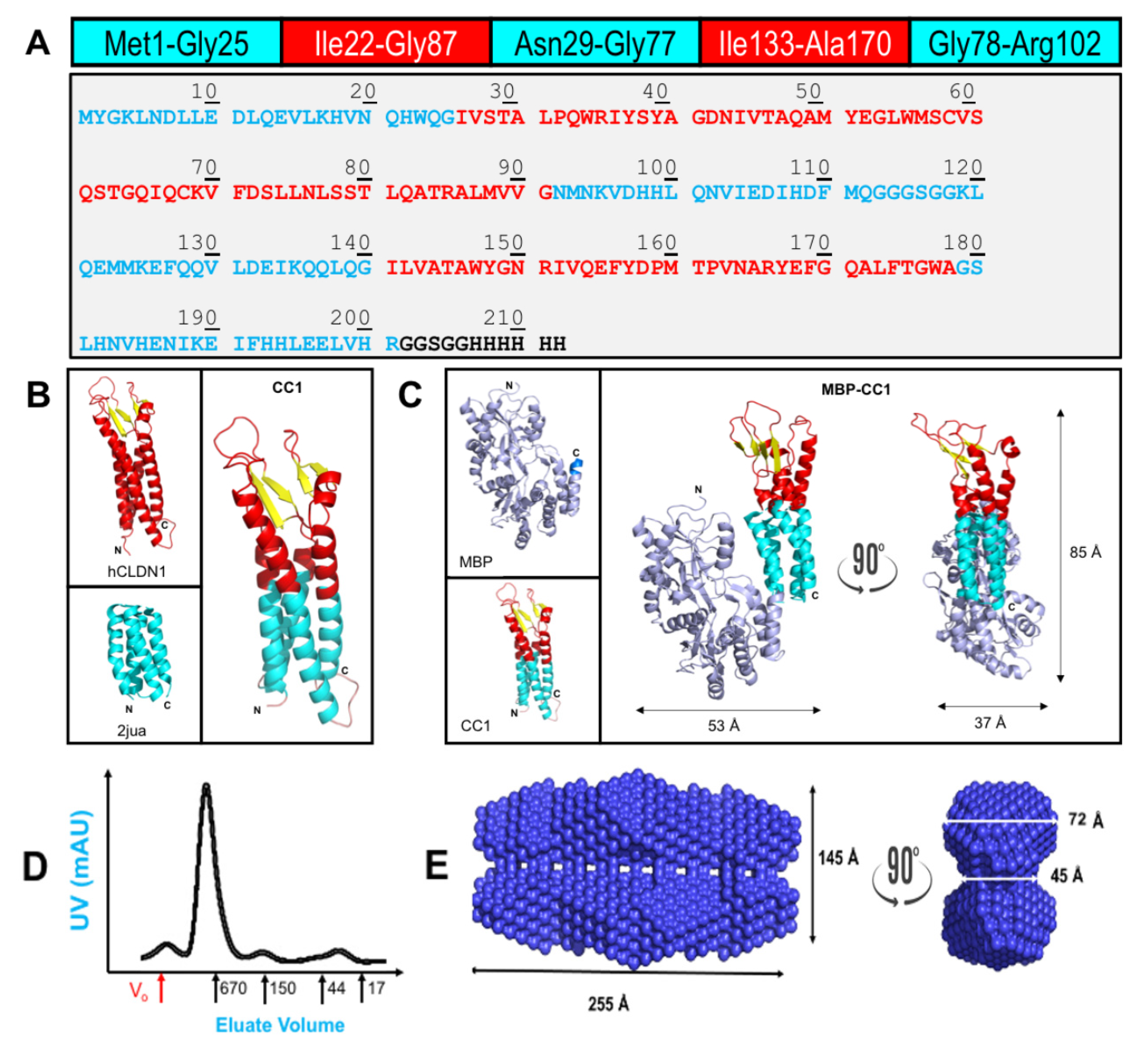
IJMS | Free Full-Text | Chimeric Claudins: A New Tool to Study Tight Junction Structure and Function
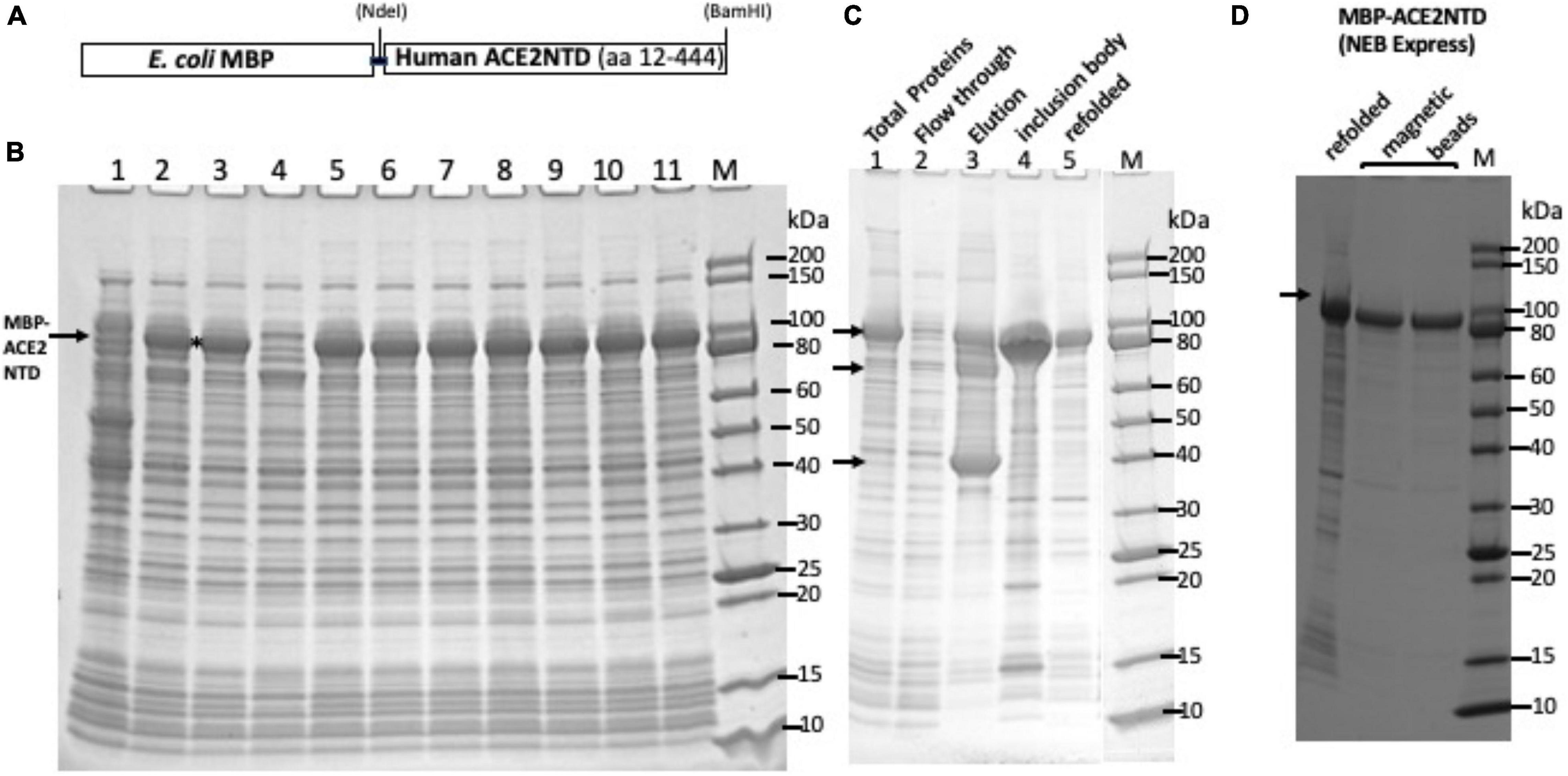
Frontiers | Expression of Human ACE2 N-terminal Domain, Part of the Receptor for SARS-CoV-2, in Fusion With Maltose-Binding Protein, E. coli Ribonuclease I and Human RNase A

Devang Mehta 🌱👨🏽🔬🧬 on X: "This week's #labhack: converting used @NEBiolabs DNA/plasmid cleanup columns into micro-spin affinity matrix columns. Tested with amylose resin, time to elution ~15mins. https://t.co/xmVHC4BPGW" / X

Expression of human ACE2 N-terminal domain, part of the receptor for SARS-CoV-2, in fusion with maltose binding protein, E. coli ribonuclease I and human RNase A | bioRxiv

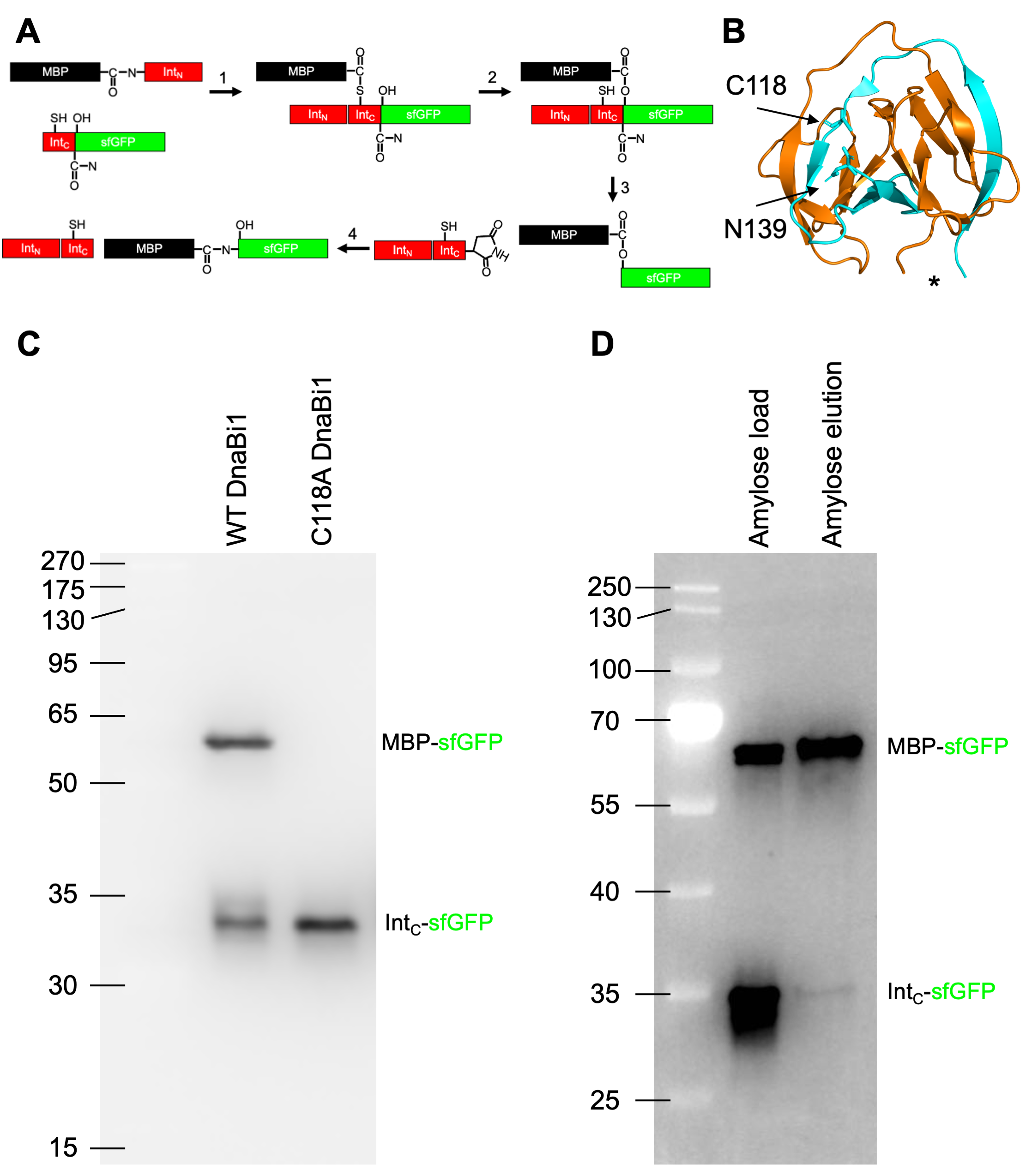
Purification of hetero-oligomeric protein variants using a modified tandem affinity purification approach - ScienceDirect


![NEB] NEBExpress® T4 Lysozyme | 코람바이오텍(주)/코람바이오젠(주) NEB] NEBExpress® T4 Lysozyme | 코람바이오텍(주)/코람바이오젠(주)](https://www.korambiotech.com/wp-content/uploads/2023/06/nebexpress-figure1-min.png)
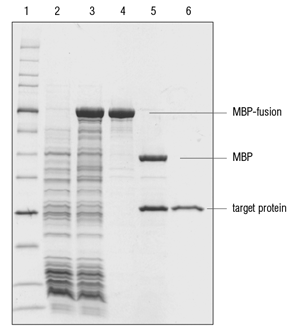
![NEB] NEBExpress® T4 Lysozyme | 코람바이오텍(주)/코람바이오젠(주) NEB] NEBExpress® T4 Lysozyme | 코람바이오텍(주)/코람바이오젠(주)](https://www.korambiotech.com/wp-content/uploads/2023/06/nebexpress-figure2-min.png)