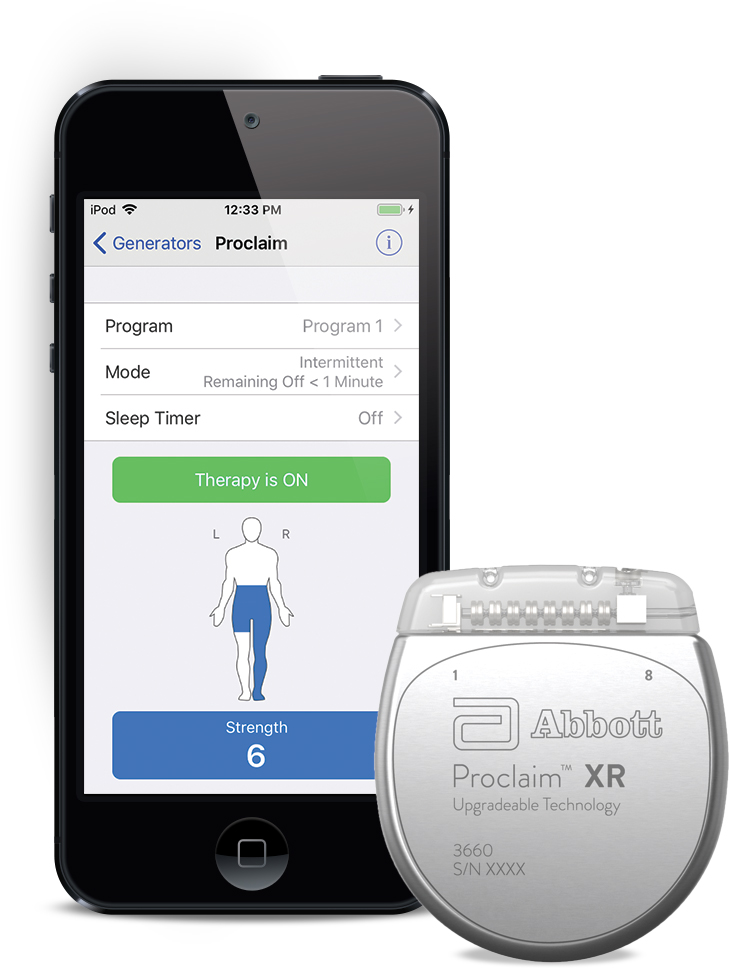
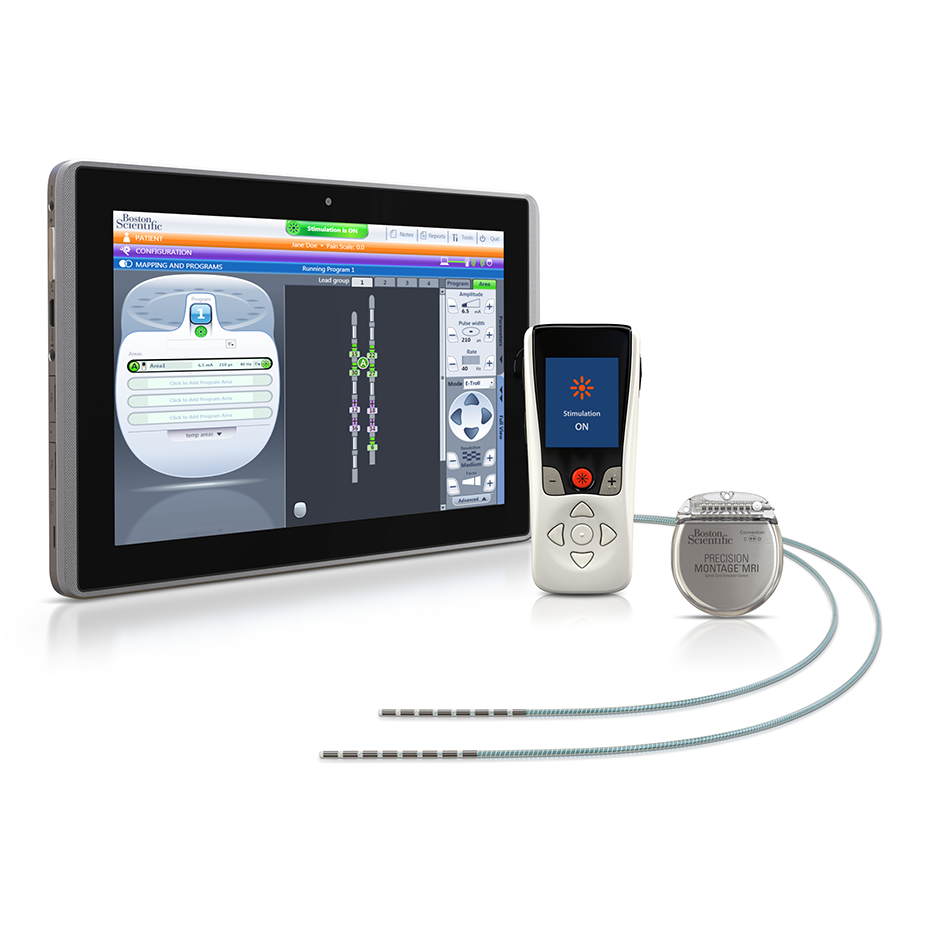
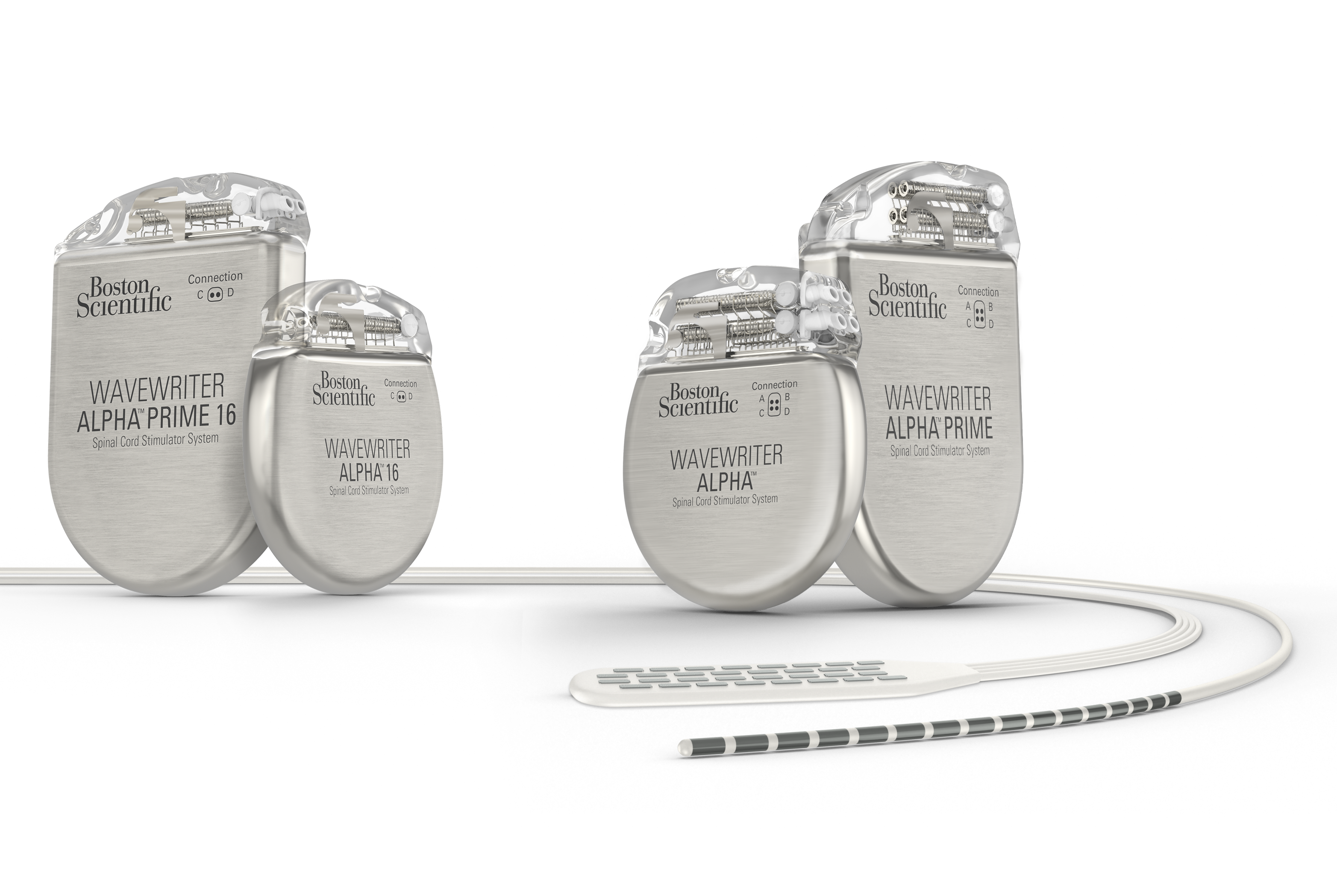
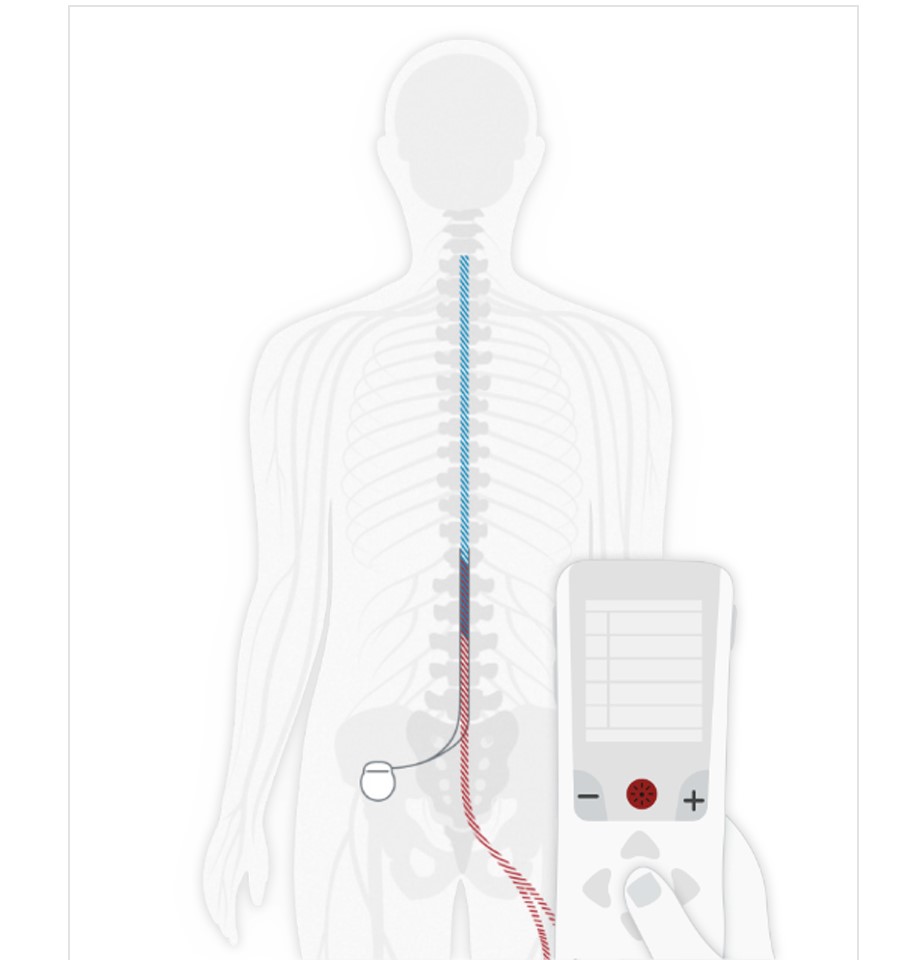
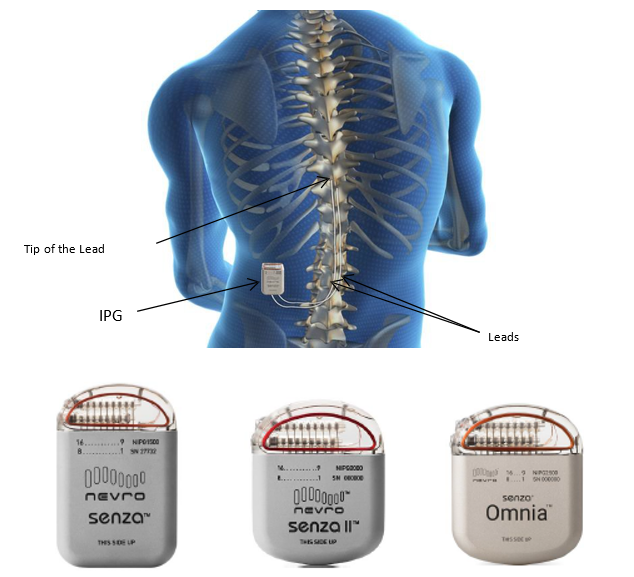
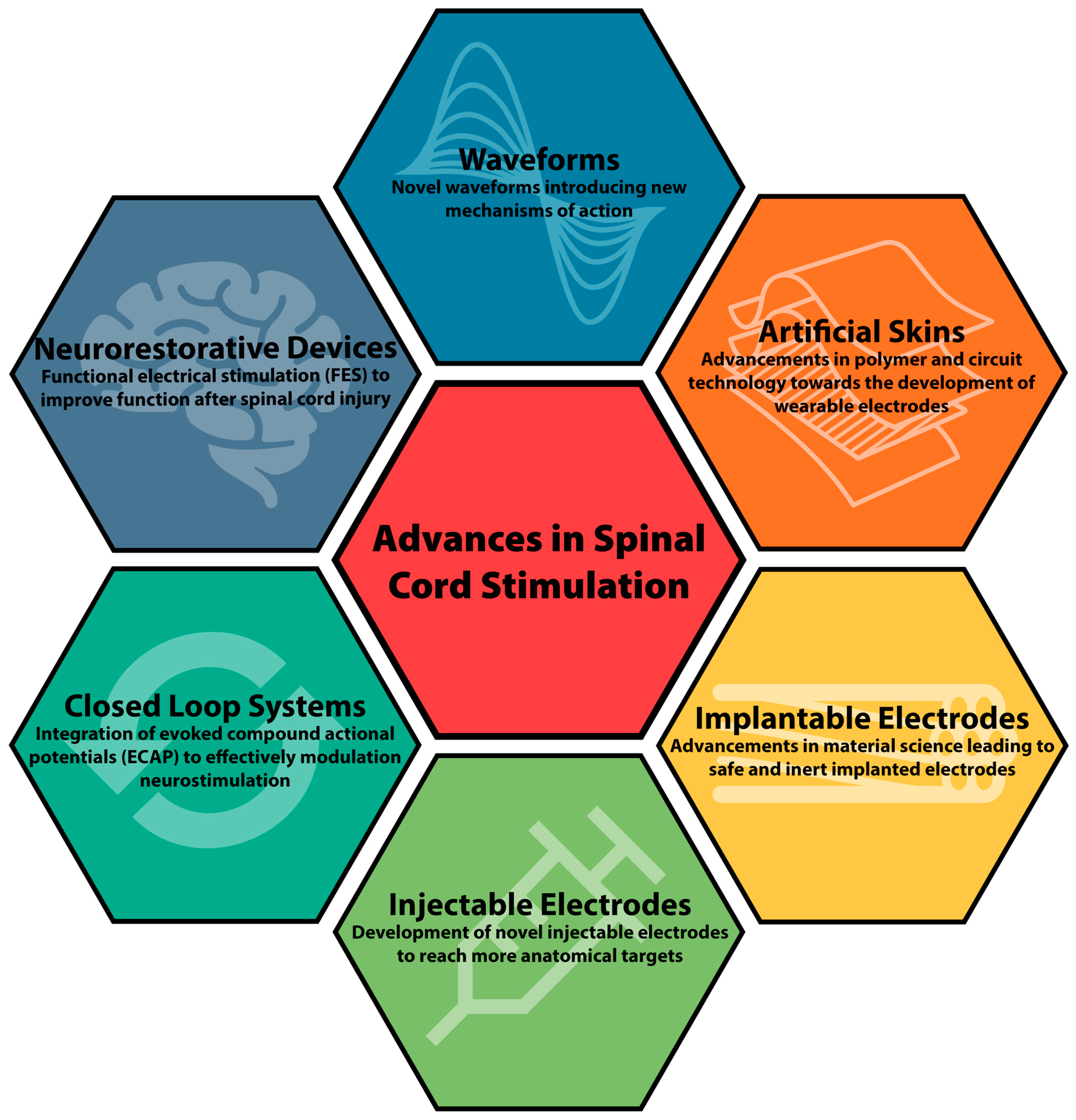
Long-term safety and efficacy of closed-loop spinal cord stimulation to treat chronic back and leg pain (Evoke): a double-blind, randomised, controlled trial - The Lancet Neurology
Medtronic Announces FDA Approval And US Launch Of Next Generation Spinal Cord Stimulator For Chronic Pain Management
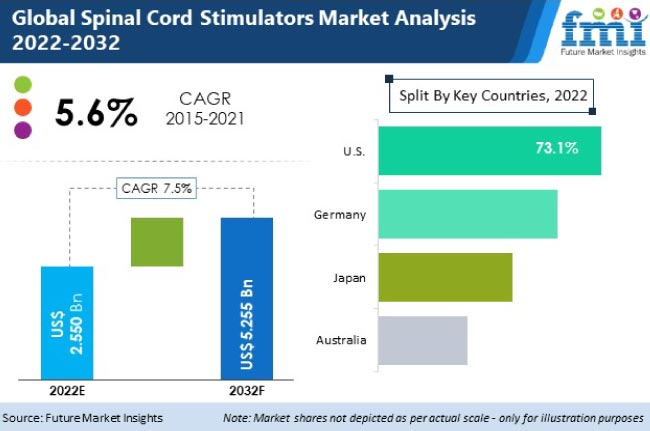
Spinal Cord Stimulators Market is expected to grow at over a CAGR of 7.5% during the forecast period (2022-2032). - PharmiWeb.com

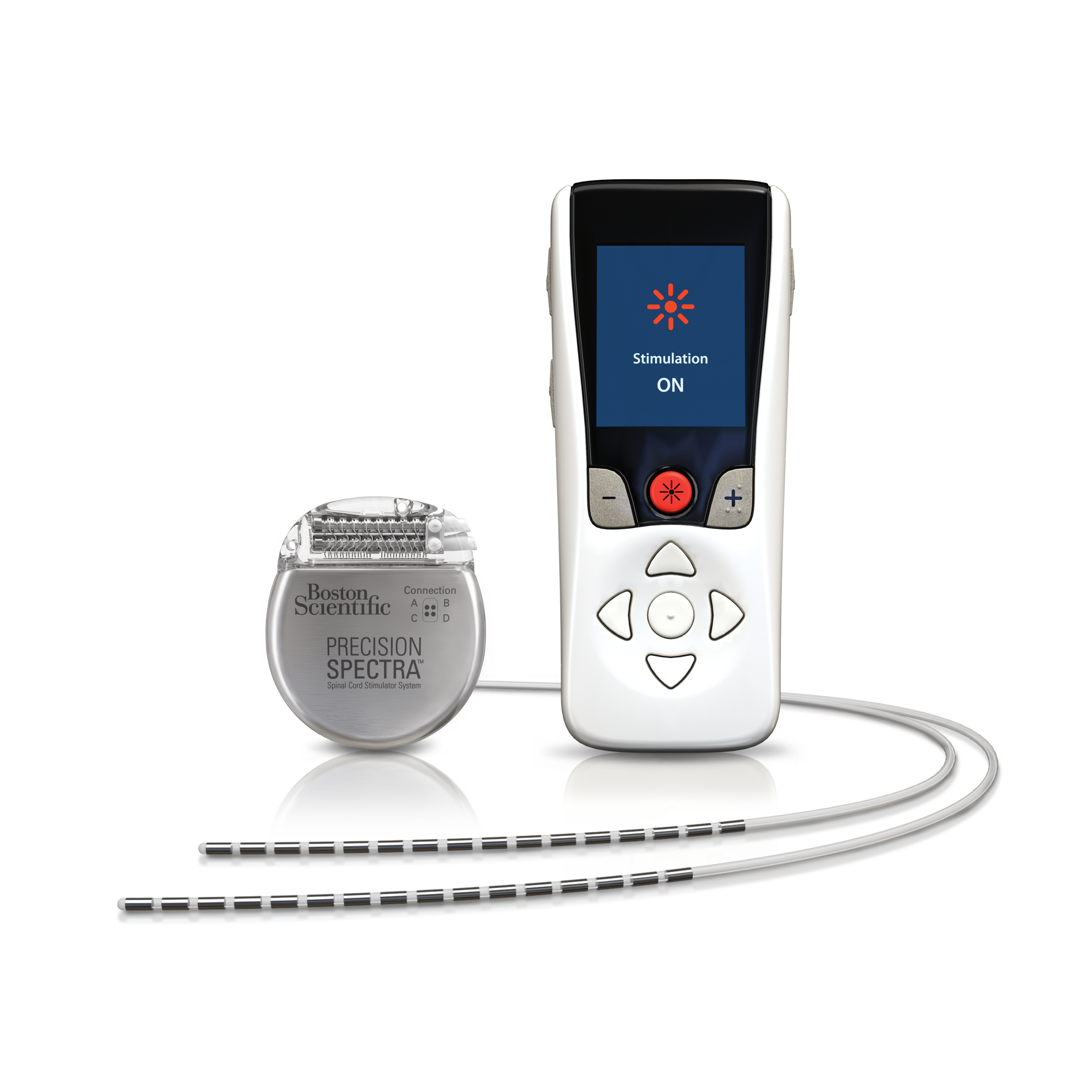
Spinal Cord Stimulator Market Top 10 companies with advanced Technologies, shares and Forecast 2020-2027
NICE recommends spinal cord stimulation system for chronic back or leg pain | The Chartered Society of Physiotherapy